本研究选取磺胺嘧啶(SDZ)作为模型污染物,该物质是一种在多种环境基质中常被检测到的磺胺类抗生素[25],旨在探究NH₄⁺对BC/PI系统中SDZ降解的影响。随后采用原位与非原位表征技术组合,包括化学淬灭实验、电子顺磁共振(EPR)光谱及电化学分析,以鉴定BC/PI系统和BC/PI/NH4+系统中的主导活性物种。此外,通过密度泛函理论(DFT)计算阐明了涉及不同活性物种的SDZ降解途径的热力学可行性。最终通过液相色谱-四极杆飞行时间质谱(LC-QTOF-MS)鉴定氧化过程中生成的硝基副产物,并运用ECOSAR预测模型评估其环境风险。
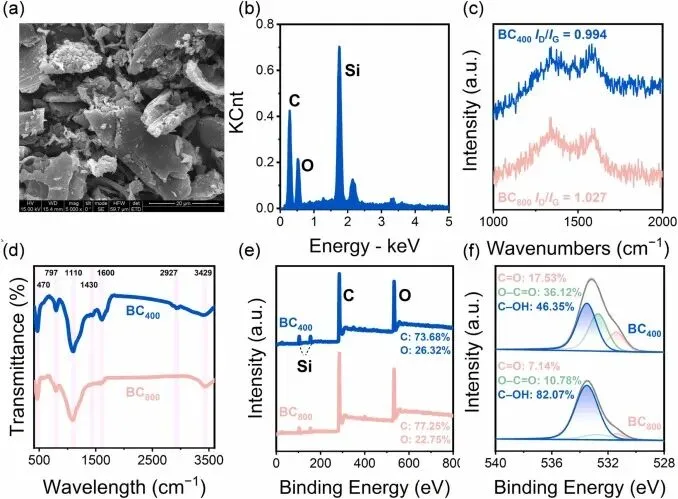
Fig. 1. (a-b) SEM image and EDX elemental mapping of BC800, (c) Raman spectra, (d) FT-IR patterns, (e) XPS survey spectra, and (f) high-resolution O 1 s XPS spectra of BC400 and BC800.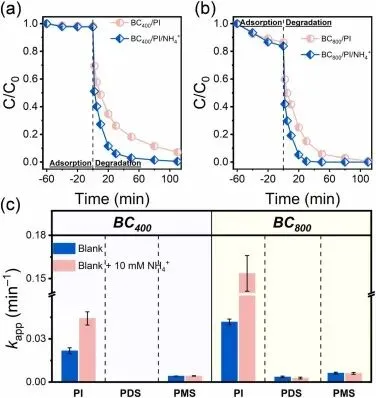
Fig. 2. Effects of NH4+ on SDZ elimination in (a) BC400/PI and (b) BC800/PI systems, (c) comparative analysis of NH4+ influence on SDZ degradation rates across different BC-AOPs systems. (Experimental conditions: [BC] = 0.2 g L−1, [SDZ] = 20 μM, [oxidants] = 0.25 mM, [NH4Cl] = 10 mM for BC/PI systems, [(NH4)2SO4] = 5 mM for BC/PDS and BC/PMS systems, initial pH: 7).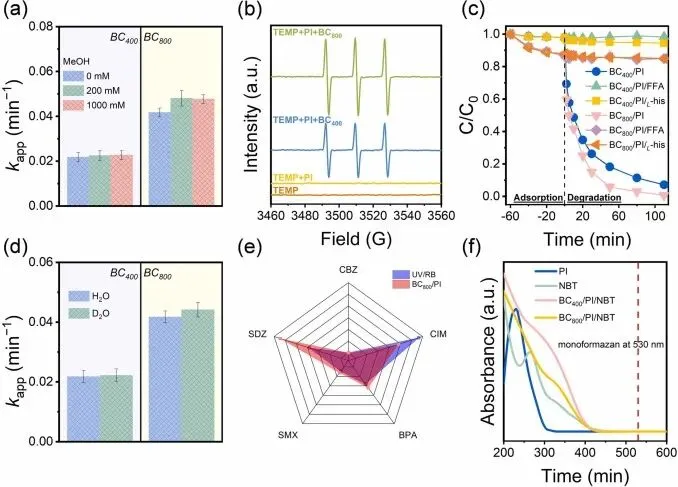
Fig. 3. (a) Effects of MeOH on the oxidation rate of SDZ in BC/PI systems, (b) EPR spectra obtained with 2,2,6,6-tetramethyl-4-piperidinol (TEMP) as a spin-trapping agent, effects of (c) FFA, L-his, or (d) solvent exchange on SDZ elimination, (e) kapp (min−1) of five organic pollutants oxidation by UV/RB and BC800/PI systems, (f) UV–vis absorption spectra of NBT in different systems (Experimental conditions: [BC] = 0.2 g L−1, [pollutants] = 20 μM (BC/PI) or 0.15 mM (UV/RB), [PI] = 0.25 mM, [TEMP] = 50 mM, [FFA] = [L-his] = 10 mM, initial pH: 7).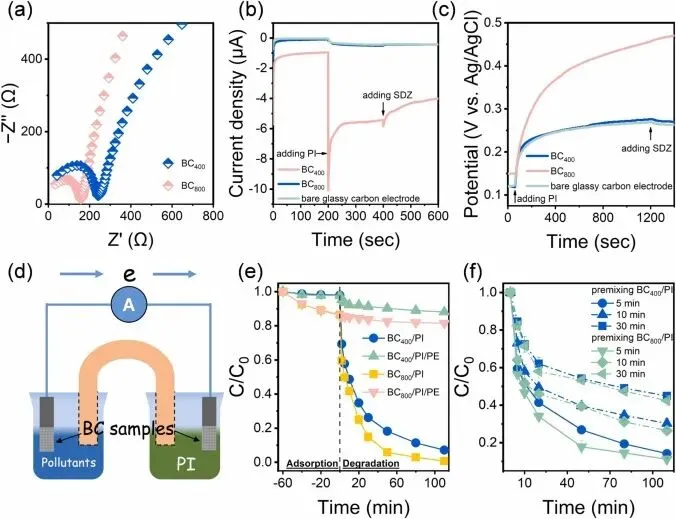
Fig. 4. (a) Electrochemical impedance characterization of BC, and (b) chronoamperometry and (c) open-circuit potential characterizations of the BC/PI systems, (d) schematic illustration of the GOP system, effects of (e) PE and (f) premixing BC and PI on SDZ elimination in BC/PI systems (Experimental conditions: [BC] = 0.2 g L−1, [SDZ] = 20 μM, [PI] = 0.25 mM, [PE] = 5 mM, initial pH: 7).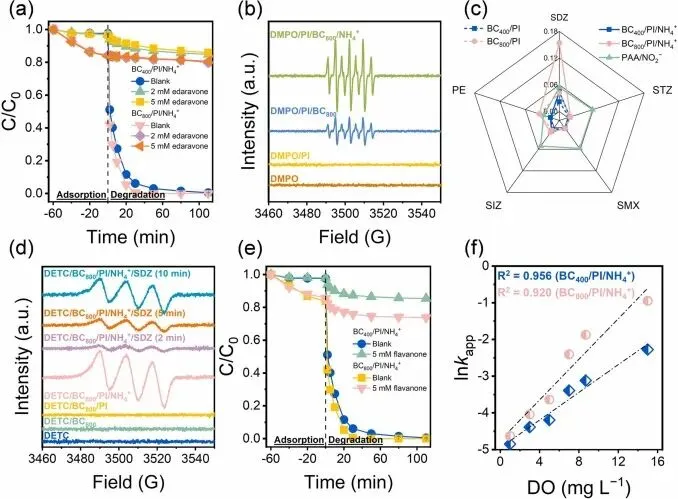
Fig. 5. (a) Effects of edaravone on SDZ elimination in BC/PI/NH4+ systems, (b) EPR spectra obtained with 5,5-Dimethyl-1-pyrroline N-oxide (DMPO) as a spin-trapping agent, (c) kapp (min−1) of five organic pollutants oxidation by PAA/NO2−, BC/PI, and BC/PI/NH4+ systems, (d) EPR spectra obtained with (DETC)2-Fe2+ as a spin-trapping agent, (e) effects of flavanone on SDZ elimination in BC/PI/NH4+ systems, (f) correlations between the DO concentration and the lnkapp in BC/PI/NH4+ systems. (Experimental conditions: [BC] = 0.2 g L−1, [pollutants] = 20 μM, [PAA] = 0.15 mM, [PI] = 0.25 mM, [DMPO] = 100 mM, [DETC] = 50 mM, [NH4+] = 10 mM, [NO2−] = 20 mM, initial pH: 7).本研究表明,NH₄⁺通过将主导活性物种从•IO₃转移至RNS(尤其是NO₂•),显著增强了BC/PI体系中的SDZ降解效率。机理研究揭示,相较于•IO3,NO2•与SDZ的反应动力学更具优势,主要源于其氧化SDZ脆弱位点(如磺酰胺氢原子和芳香氮原子)的能量障碍更低。然而,NO2•介导的反应促进了硝基副产物形成,液相色谱-四极杆飞行时间质谱(LC-QTOF-MS)鉴定出硝基酚衍生物。毒性预测表明,多种硝基中间体展现出高于母体SDZ的急性与慢性生态毒性。研究揭示了NH₄⁺在SDZ降解过程中BC/PI体系中的双重特性:既增强了污染物降解效率,又通过毒性副产物形成意外提升了生态风险。这强调了实施PI-AOP工艺时需审慎评估共存无机离子的必要性。本研究局限在于缺乏硝基副产物(如硝基酚类)的定量分析,导致环境风险评估不够精准。后续研究可聚焦于量化这些有毒中间体,以更准确评估BC/PI系统在废水处理中的实际应用价值。
Jing Dai, Ning Chen, Yujun Wang, Xianghao Zha, Juan Wang, Ziyan Yang, Guodong Fang, Reactive nitrogen species regulating sulfadiazine degradation by biochar-activated periodate with ammonium, Applied Catalysis B: Environment and Energy, 2026, https://doi.org/10.1016/j.apcatb.2025.125979
声明:本公众号仅分享前沿学术成果,无商业用途。如涉及侵权,请立刻联系公众号后台或发送邮件,我们将及时修改或删除!
邮箱:Environ2022@163.com