在本研究中,我们提出了一种利用共价键共享的CoOh-O-MnOh构型来诱导Co-Mn反尖晶石结构中d电子重新分布的策略,旨在实现体相稳定性与高PMS活化性能之间的平衡。通过理论计算和同步辐射X射线吸收光谱(XAS)分析,证实了CoOh-O-MnOh构型的存在及其伴随的电子重分布现象。结果表明,通过优化 CoOh-O-MnOh 构型,Co-Mn 反尖晶石的性能显著提升。在相对较低的催化剂(0.2 g/L)和 PMS(0.5 mM)用量下,它可以在 15 分钟内实现 20 mg/L BPA 的 100% 去除率。表观速率常数达到0.389 min⁻¹,是未经改性的Co₃O₄(k_(obs)Co₃O₄=0.029 min⁻¹)的13.4倍。这种增强归因于 CoMn2O4 优化后的 O 2p 带中心,其中氧作为“电子桥”,在 Co 和 Mn 的协同作用下促进了更快的电荷平衡。当前的电子环境有利于非自由基主导的途径,这些途径主要涉及 CoIV=O 和 1O2。该体系最大限度地发挥了反尖晶石结构的潜力,并提高了对阴离子的耐受性(在 100 mM 背景阴离子条件下,平均效率偏差小于 6.5%)。随后的毒性评估表明,其降解产物对环境无害。本文概述的方法有助于精确调节催化活性和反应途径,从而为设计适用于 AOP 的稳健、抗干扰催化剂提供了新的范式。
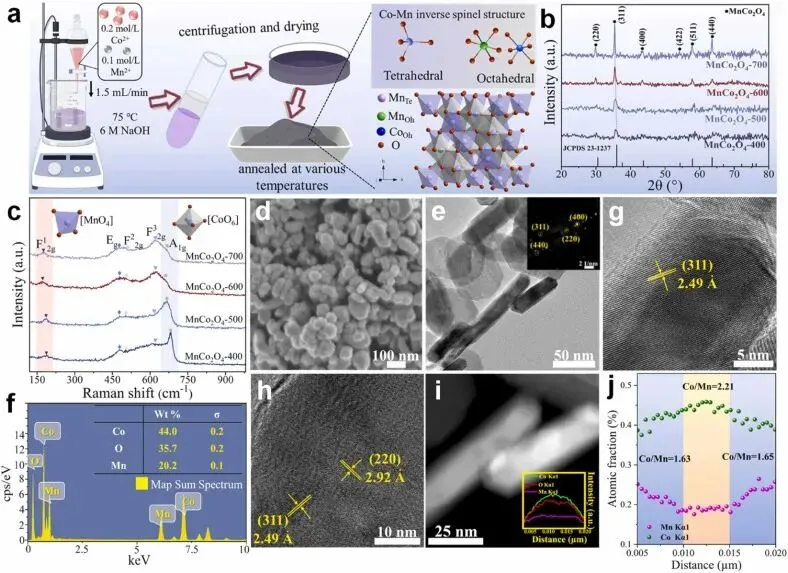
Fig. 1. (a) The synthetic procedure, (b) XRD patterns, and (c) Raman spectra of MnCo2O4-x (x = 400,500,600,700); (d) SEM image, (e) TEM image, (f) EDS spectrum, and (g-h) HR-TEM images of MnCo2O4-600; (i) EDS line scan across the particle along the arrows image for MnCo2O4-600 and (j) atomic ratio of Co and Mn obtained from the EDS line scan across the particle along the arrows in (i).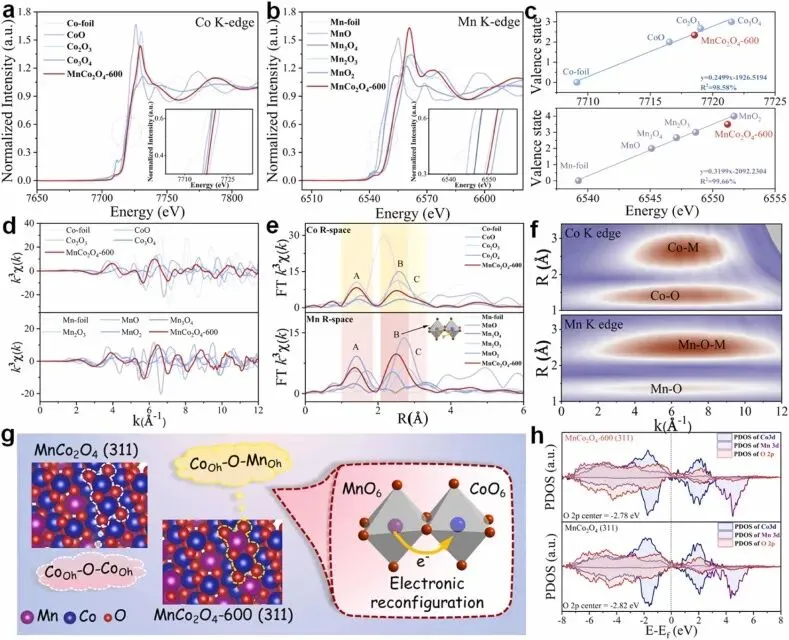
Fig. 2. XAS analyses. XANES spectra of the (a) Co and (b) Mn K-edge; (c) average oxidation states of Co and Mn in samples/standards obtained from the XANES spectra; (d) κ3-weighted EXAFS spectra and (e) corresponding FT-EXAFS spectra of Co and Mn at the R-space; (f) WTs of the Co and Mn K-edges of the EXAFS spectral profiles of the MnCo2O4-600; (g) structural diagram of the surface coordination structure; (h) PDOSs of Co 3d, Mn 3d, and O 2p orbitals of the MnCo2O4 and MnCo2O4-600 surfaces, respectively.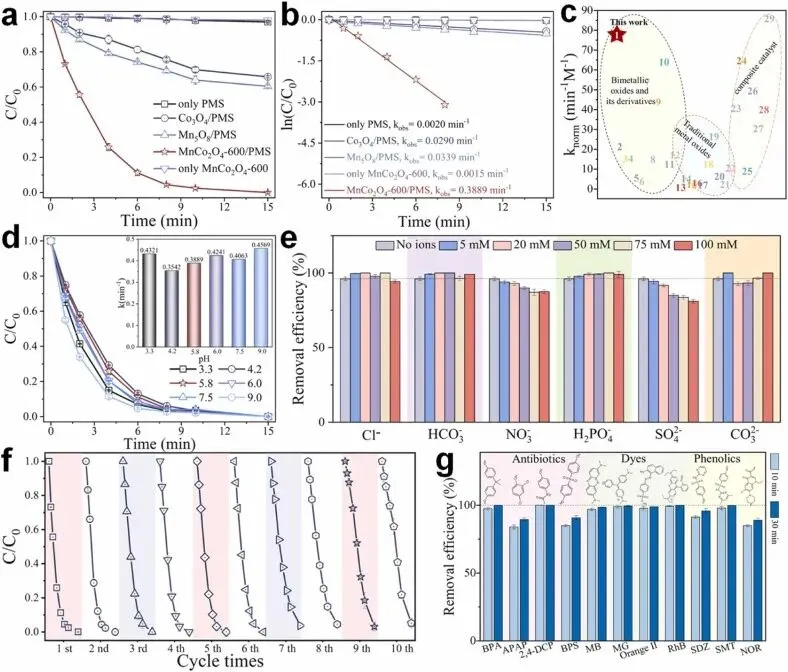
Fig. 3. (a) Catalytic performance of the as-synthesized materials and (b) corresponding kinetic constants; (c) normalized k comparison with other systems; (d) BPA degradation performance in MnCo2O4-600/PMS system under different initial pH conditions; (e) effects of common anions in the MnCo2O4-600/PMS system on BPA degradation; (f) reusability of MnCo2O4-600; and (g) removal efficiency after 10 min and 30 min reaction of different pollutants in MnCo2O4-600/PMS system. Experimental conditions: [catalyst] = 0.2 g/L, [BPA] = 20 mg/L, [PMS] = 0.5 mM, initial pH = 5.8, T = 298 K.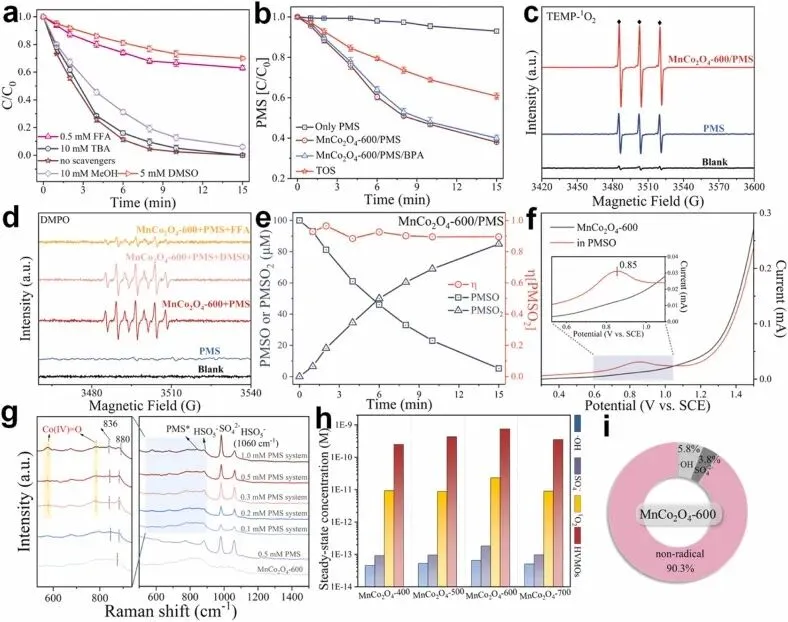
Fig. 4. Roles of the reactive species in the MnCo2O4-600/PMS system. (a) Quenching experiments of the MnCo2O4-600/PMS system; (b) abatement of PMS in the solution and TOS in the MnCo2O4-600/PMS system; (c) EPR spectra of the different systems captured by TEMP; (d) EPR spectra of the different systems captured by DMPO; (e) degradation of PMSO and production of PMSO2 during the MnCo2O4-600/PMS system; (f) LSV measurements of MnCo2O4-600 with different contents; (g) in-situ Raman spectra views in the MnCo2O4-600/PMS system; (h) the steady-state concentrations of free radicals or nonradicals and (i) their proportions.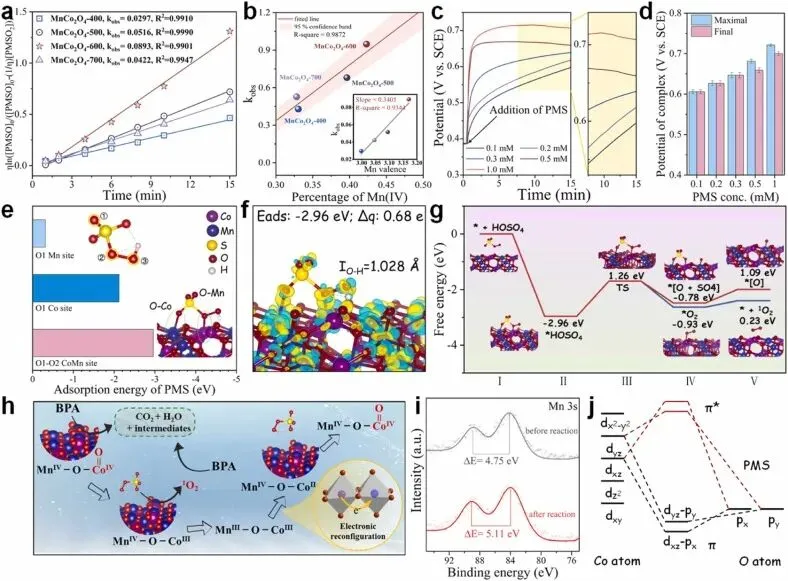
Fig. 5. (a) Plot of versus time in various systems; (b) correlation of the percentage of MnⅣ in various systems of kobs corresponding to (a); (c) CP measurements on MnCo2O4-600; (d) maximum and final potentials of the complex within 900 s on MnCo2O4-600; (e) adsorption ability of PMS at different sites on the material surface; (f) charge density difference in a PMS molecule adsorbed on MnCo2O4-600; (g) MnCo2O4-600 forms a reaction energy barrier for CoIV=O and 1O2; (h) schematic representation of the reaction process between nonradical and BPA; (i) HR-XPS spectra of Mn 3 s of MnCo2O4-600 before and after reaction; and (j) schematic representation of bonding information for the Co=O portion of CoIV=O.总而言之,我们阐明了一种基本机制:通过对Co-Mn反尖晶石中边共享的CoOh-O-MnOh构型进行位点特异性设计,可增强PMS的活化作用,并促进污染物降解的高效非自由基途径。通过共沉淀-煅烧法精确构建八面体配位环境,我们证明调节共边相互作用可诱导八面体间的电子重分布,这不仅稳定了MnIV并抑制了JT畸变,还提高了活性CoII的浓度。这种电子重分布优化了O 2p能带中心,加速了电子转移动力学,并建立了一座促进电荷平衡的电子桥。因此,重构后的电子结构最大限度地发挥了催化剂的固有潜力,主要通过CoIV=O和1O2途径驱动非自由基氧化反应。得益于坚固八面体框架内动态的价态再平衡,这种优化的尖晶石还表现出卓越的稳定性和对背景阴离子的耐受性。八面体Co和Mn位点之间的相互作用,通过自旋态调制和共价电子桥接最大限度地发挥了反尖晶石结构的潜力,这为设计抗干扰的AOP催化剂提供了一种新策略。
Qi Zhang, Wei Li, Siyuan Cao, Xu Yin, Hongwei Zhu, Liankai Gu, Haoming Chen, Xiaoyuan Zhang, Weiqing Han, Kajia Wei, CoOh-O-MnOh edge-sharing engineering in Co-Mn inverse spinel oxides for PMS activation: Electronic reconfiguration-driven nonradical oxidation via synergistic CoⅣ=O and 1O2, Applied Catalysis B: Environment and Energy, 2026, https://doi.org/10.1016/j.apcatb.2025.126149
声明:本公众号仅分享前沿学术成果,无商业用途。如涉及侵权,请立刻联系公众号后台或发送邮件,我们将及时修改或删除!
邮箱:Environ2022@163.com