南京工业大学罗宏斌/加州州立大学Liu Yangyang/南开大学张振杰合作Angew: 两性离子“赋能”乙烯基COF,构建高性能质子电解质
- 2026-05-23 06:41:45


第一作者:李浩宇、张国芹
通讯作者:罗宏斌、Yangyang Liu、张振杰
通讯单位:南京工业大学、加州州立大学洛杉矶分校、南开大学
近日,南京工业大学罗宏斌、加州州立大学Liu Yangyang、南开大学张振杰教授团队合作,在固态质子电解质领域取得重大突破,研究成果发表在国际顶级期刊《Angewandte Chemie International Edition》上。通过在磺基甜菜碱两性离子乙烯基连接共价有机框架(ZVCOF)孔道中负载磷酸(PA),研究团队制备获得了复合固态质子电解质PA@ZVCOF。该电解质在常温下的质子传导率高达5.34 × 10-2 S cm-1,创造了COF基质子电解质在常温下的新纪录。同时,其高温质子传导率(1.79 × 10-1 S cm-1)也处于目前最先进水平。基于此电解质组装的固态质子电池,展现出108.5 mAh g-1的高比容量(1.0 A g-1电流密度)和卓越的长循环稳定性(循环2000次后容量保持率高达90%),两项关键性能指标均刷新了固态质子电池的记录。尤为重要的是,该固态电解质有效解决了传统液态酸电解质腐蚀电极的难题,为开发高安全、长寿命的下一代储能器件提供了全新方案。
随着对能源安全和能量密度的要求日益提高,开发高性能、高安全性的下一代电池技术成为科研界和产业界的焦点。质子电池因其电荷载体质子(H+)具有最小的离子半径、最低的原子质量和丰富的自然资源,展现出优异的倍率性能和成本优势,被视为极具潜力的新型储能系统。然而,质子电池中传统酸性电解液面临易泄露、电化学窗口窄、对电极材料溶解以及电池组件腐蚀等挑战。因此,开发高性能的固态质子电解质是推动质子电池走向实用的关键。在众多固态电解质材料中,COFs因其结构可调、孔道规整、化学稳定性好等优点,成为理想的候选平台。但现今COF基质子导体大多需要在高温或高湿环境下才能实现高质子传导率,在室温无外加湿度下的性能普遍不佳,这极大地限制了其在质子电池中的应用。
针对上述挑战,研究团队独辟蹊径,提出了一种“两性离子工程”设计策略,在COF骨架中引入兼具正负电荷基团的两性离子官能团,旨在协同解决质子解离与质子迁移这两个关键科学问题。引入的两性离子官能团在COF骨架中形成一个个“微型质子泵”,有效促进了质子的解离与迁移过程。实验结果表明,基于两性离子COF的复合电解质在室温下的质子传导率高达5.34 × 10-2 S cm-1,成功突破了COF基质子导体在室温无外加湿度条件下的性能瓶颈,展现出与商业Nafion膜相当甚至更优的传导性能。此外,将该电解质应用于固态质子电池中,不仅实现了创纪录的比容量和优异的循环稳定性,还直观证明了固态电解质对电极的有效保护作用,为替代传统液态酸电解质提供了有力支撑。
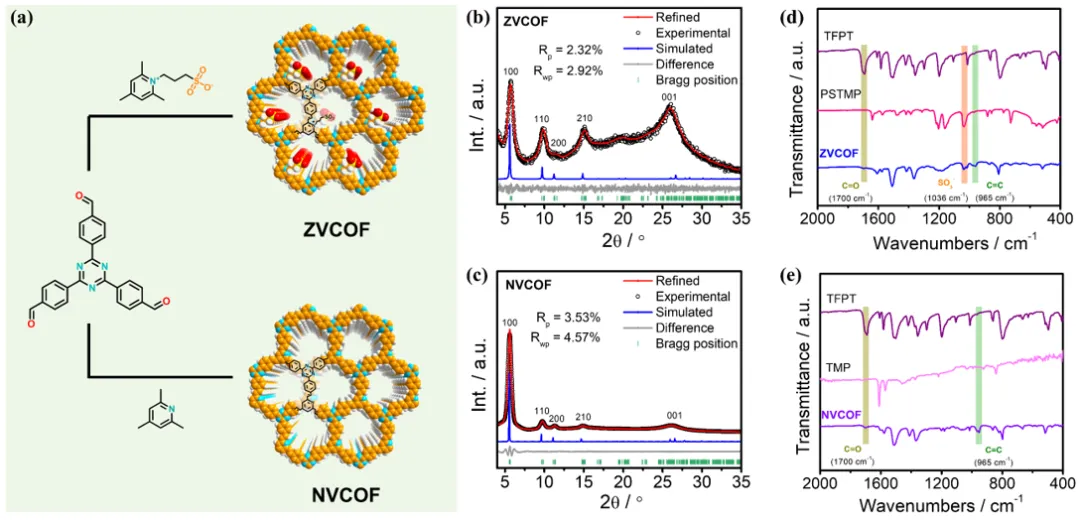
Figure 1. (a) Schematic syntheses and (b, c) Pawley refinement of PXRD patterns for ZVCOF and NVCOF. (d, e) FT-IR spectra of ZVCOF, NVCOF, and corresponding monomers.
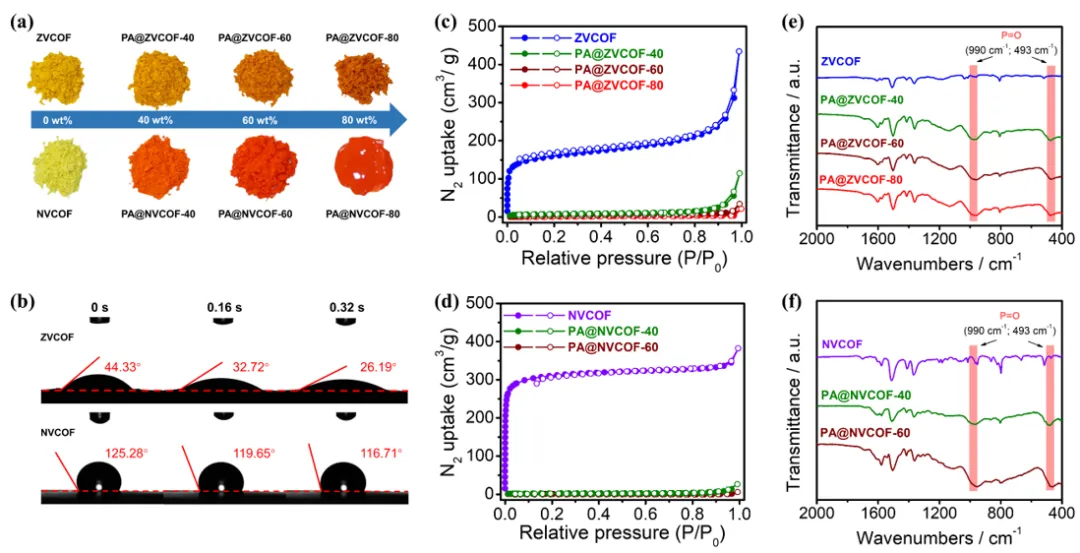
Figure 2. (a) Optical images of ZVCOF, NVCOF, and the corresponding composites with different PA loading. (b) Time-dependent contact angles of PA on the surfaces of ZVCOF and NVCOF pellets. (c, d) N2 adsorption-desorption isotherms at 77 K and (e, f) FT-IR spectra of ZVCOF, NVCOF, and the corresponding composites with different PA loading.
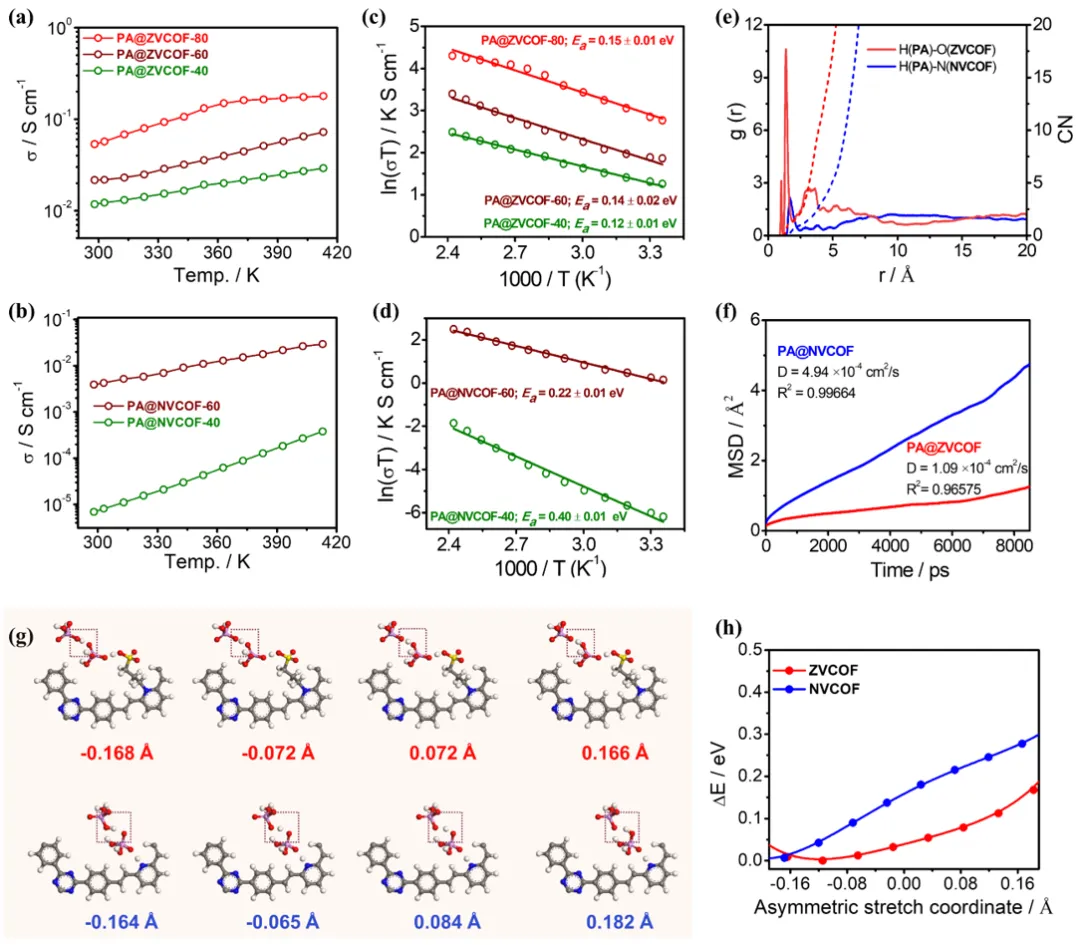
Figure 3. (a, b) Temperature-dependent proton conductivity and (c, d) Arrhenius plots of the proton conductivities of PA@ZVCOF-X (X = 40–80) and PA@NVCOF-X (X = 40–60). (e) Radial distribution functions (RDF) of H(PA)-O(ZVCOF) and H(PA)-O(NVCOF), along with the coordination numbers (CN). (f) Calculated mean square displacement (MSD) of PA molecules as a function of the simulation time. (g) Simulated snapshots illustrating the proton dissociation process of a representative PA molecule within the ZVCOF and NVCOF frameworks, along with (h) the corresponding relative energy profiles.
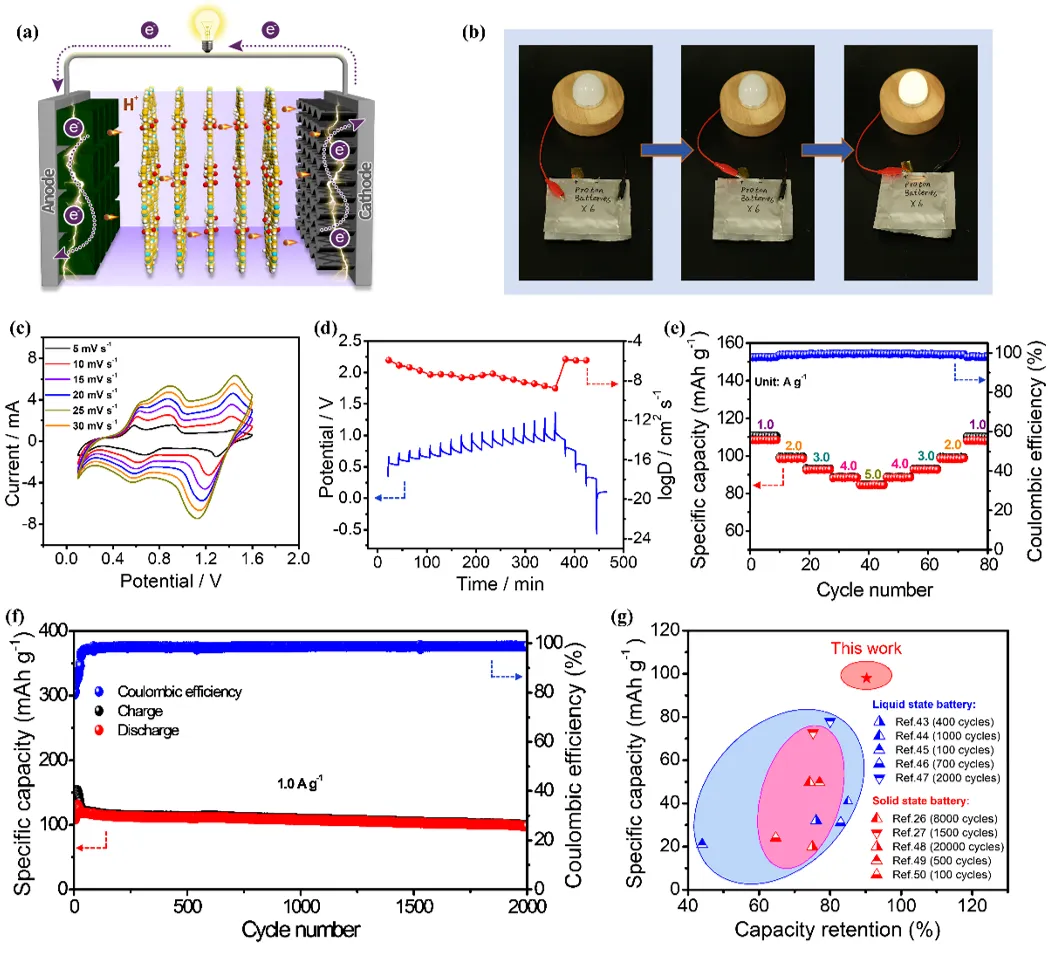
Figure 4. (a) Schematic representation of a solid-state proton battery using PA@ZVCOF-80 electrolyte. (b) Photographs of an LED bulb lighted up by the solid-state proton battery. (c) CV curves at different scan rates, (d) GITT curve and corresponding proton diffusion coefficient of the solid-state proton battery. (e) Rate performance of the solid-state proton battery. (f) Cycling stability and Coulombic efficiency of the solid-state proton battery at a current density of 1.0 A g-1. (g) Comparison of specific capacity and capacity retention of the solid-state proton battery using PA@ZVCOF-80 electrolyte with other reported proton batteries.
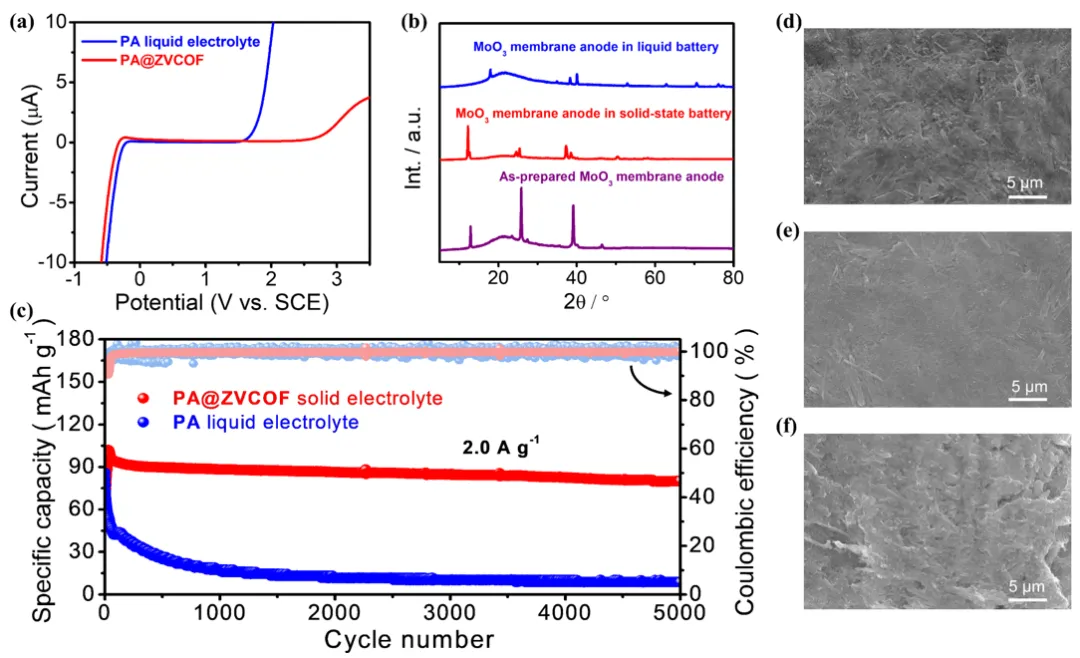
Figure 5. (a) Linear scanning voltammetry curves of PA@ZVCOF-80 and a PA aqueous electrolyte (5.0 M). (b) XRD patterns of the as-prepared MoO3 membrane anode (purple), the MoO3 membrane anode after cycling tests in a solid-state proton battery (red) and in a proton battery with PA aqueous electrolyte (5.0 M; blue). (c) Cycling performance of proton batteries using PA@ZVCOF-80 or a PA aqueous electrolyte at the current density of 2.0 A g-1. SEM images of (d) as-prepared MoO3 membrane anode, the MoO3membrane anode after cycling tests in (e) solid-state proton battery and (f) proton battery with PA aqueous electrolyte.
Hao-Yu Li, Guo-Qin Zhang, Hong-Bin Luo,* Jin Zhang, Dong-Sheng Shao, Qiao Qiao, Fengdong Wang, Yangyang Liu,* Zhenjie Zhang,* Xiao-Ming Ren, Zwitterionic Engineering of Vinylene-Linked Covalent Organic Frameworks for Superior Protonic Electrolytes. Angew. Chem. Int. Ed. 2026, e7677012. doi.org/10.1002/anie.7677012.
版权声明
测试表征+计算+绘图

