南京大学鲁艺/孙为银/斯克利普斯研究所余金权JACS Au:通过SET和CMD途径实现吲哚啉的C5和C7位硫化反应
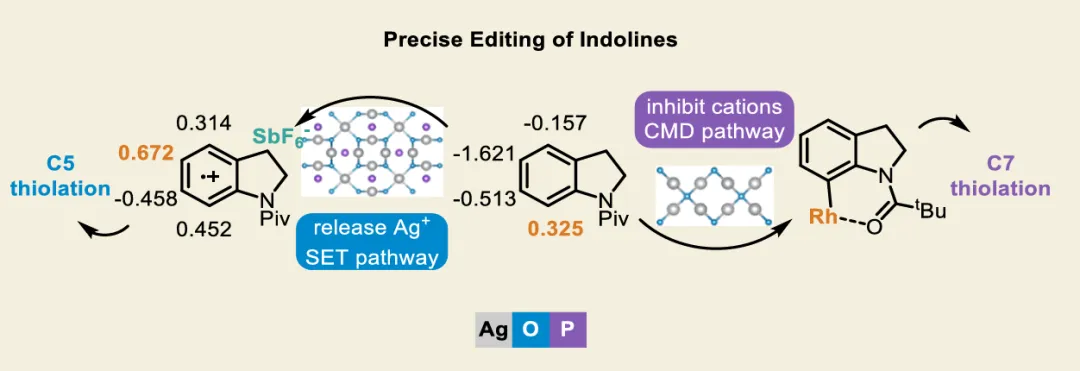 In drug discovery, the pursuit of concise and specific synthetic methods is driving the development of new strategies. Particularly during the latestages of a synthetic sequence, when the substrate molecule contains multiple highly similar C−H bonds, the ability to achieve selective “molecular editing” using mild and precise transformations has become a crucial capability. Here, we report Rh-catalyzed molecular editing of indoline derivatives at different positions via a single, oxidant-tunable catalytic system. Using Ag3PO4 as the oxidant, Rh promotes the formation of indoline radical cations stabilized by SbF6−, enabling remote C5-thiolation via a single-electron transfer (SET) mechanism, as supported by the Fukui function. In contrast, Ag2O suppresses the SET pathway, as confirmed by the ABTS assay, and enables C7-selective thiolation through a concerted metalation−deprotonation (CMD) process mediated by a Rh(III)/MPAA ligand system. Mechanistic experiments and Fukui indices calculation elucidate the origin of the observed regiodivergence, highlighting the dual reactivity of Rh(III) and offering new conceptual insights into oxidant-controlled C−H functionalization.图2. 研究背景及本策略 (图片来源于JACS Au)图3. 吲哚啉的位点选择性硫化反应条件优化 (图片来源于JACS Au)图4. 通过自由基途径实现吲哚啉C5硫化反应底物范围 (图片来源于JACS Au)图5. 通过CMD途径实现吲哚啉C7硫化反应底物范围 (图片来源于JACS Au)图6. 通过两种途径实现含不同导向基的吲哚啉硫化反应 (图片来源于JACS Au)图7. 克级合成和合成应用 (图片来源于JACS Au)图9. 可能的反应机理 (图片来源于JACS Au)In summary, we have discovered a pathway-controlled Rh(III) catalyzed C5-/C7-selective inert C−H thiolation for indoline editing. The Rh(III) catalyst exhibits dual reactivity by enabling site-selective C−H functionalization via distinct mechanistic pathways. Comprehensive mechanism investigations have elucidated the underlying principles governing each pathway, thereby offering a cohesive rationale that builds upon previous studies of C−H thiolation chemistry. This work not only advances the understanding of regioselective C−H functionalization but also provides valuable insights for designing future strategies in remote site-selective thiolation. Furthermore, both C5 and C7 thiolation reactions exhibit broad functional group tolerance and maintain a high efficiency on a gram scale, highlighting their potential applicability in industrial synthesis.
In drug discovery, the pursuit of concise and specific synthetic methods is driving the development of new strategies. Particularly during the latestages of a synthetic sequence, when the substrate molecule contains multiple highly similar C−H bonds, the ability to achieve selective “molecular editing” using mild and precise transformations has become a crucial capability. Here, we report Rh-catalyzed molecular editing of indoline derivatives at different positions via a single, oxidant-tunable catalytic system. Using Ag3PO4 as the oxidant, Rh promotes the formation of indoline radical cations stabilized by SbF6−, enabling remote C5-thiolation via a single-electron transfer (SET) mechanism, as supported by the Fukui function. In contrast, Ag2O suppresses the SET pathway, as confirmed by the ABTS assay, and enables C7-selective thiolation through a concerted metalation−deprotonation (CMD) process mediated by a Rh(III)/MPAA ligand system. Mechanistic experiments and Fukui indices calculation elucidate the origin of the observed regiodivergence, highlighting the dual reactivity of Rh(III) and offering new conceptual insights into oxidant-controlled C−H functionalization.图2. 研究背景及本策略 (图片来源于JACS Au)图3. 吲哚啉的位点选择性硫化反应条件优化 (图片来源于JACS Au)图4. 通过自由基途径实现吲哚啉C5硫化反应底物范围 (图片来源于JACS Au)图5. 通过CMD途径实现吲哚啉C7硫化反应底物范围 (图片来源于JACS Au)图6. 通过两种途径实现含不同导向基的吲哚啉硫化反应 (图片来源于JACS Au)图7. 克级合成和合成应用 (图片来源于JACS Au)图9. 可能的反应机理 (图片来源于JACS Au)In summary, we have discovered a pathway-controlled Rh(III) catalyzed C5-/C7-selective inert C−H thiolation for indoline editing. The Rh(III) catalyst exhibits dual reactivity by enabling site-selective C−H functionalization via distinct mechanistic pathways. Comprehensive mechanism investigations have elucidated the underlying principles governing each pathway, thereby offering a cohesive rationale that builds upon previous studies of C−H thiolation chemistry. This work not only advances the understanding of regioselective C−H functionalization but also provides valuable insights for designing future strategies in remote site-selective thiolation. Furthermore, both C5 and C7 thiolation reactions exhibit broad functional group tolerance and maintain a high efficiency on a gram scale, highlighting their potential applicability in industrial synthesis.