西安交大师仁义/武大雷爱文/南京理工王鹏程ACS Catal.:镍催化通过水氧化的电化学交叉亲电偶联反应
图1. TOC (图片来源于ACS Catal.)Nickel-catalyzed cross-electrophile coupling (XEC) has emerged as an efficient and economical strategy for constructing C−C bonds, a pivotal transformation in diversifying molecular architectures. However, conventional XEC methodologies typically rely on stoichiometric metallic reductants, which present inherent challenges, including safety risks, operational instability, and environmental concerns. Although electrochemical XEC in undivided cells circumvents the need for chemical reductants, it remains constrained by environmental issues and chemoselectivity limitations due to its dependence on sacrificial metal anodes or stoichiometric organic donors to supply electrons for cathodic reduction. Herein, we report a nickel-catalyzed electrochemical cross-electrophile coupling paired with water oxidation. By utilizing water as a sacrificial electron donor, this electrochemical platform facilitates the versatile construction of diverse C−C bonds, including Csp2−Csp3, Csp3−Csp3, and Csp−Csp3 linkages, from readily accessible aryl, alkenyl, alkynyl, and alkyl halide electrophiles, affording products in yields up to 99%. The undivided cell configuration markedly reduces system complexity, lowers capital costs, and supports scalable electrochemical synthesis. Moreover, this electroreductive coupling strategy exhibits broad functional group tolerance and is amenable to the late-stage derivatization of complex drugs and natural products. This operationally simple, electricity-driven approach offers a versatile platform for C−C bond formation.图2. 研究背景及本策略 (图片来源于ACS Catal.)图3. 条件优化 (图片来源于ACS Catal.)图4. 循环伏安实验 (图片来源于ACS Catal.)图5. 芳基碘和烷基碘的电化学交叉亲电偶联反应底物拓展 (图片来源于ACS Catal.)图6. 电化学交叉亲电偶联反应构建Csp/sp2/sp3-Csp3键底物拓展及生物相关活性分子的后期修饰反应 (图片来源于ACS Catal.)图7. 放大反应、控制实验 (图片来源于ACS Catal.)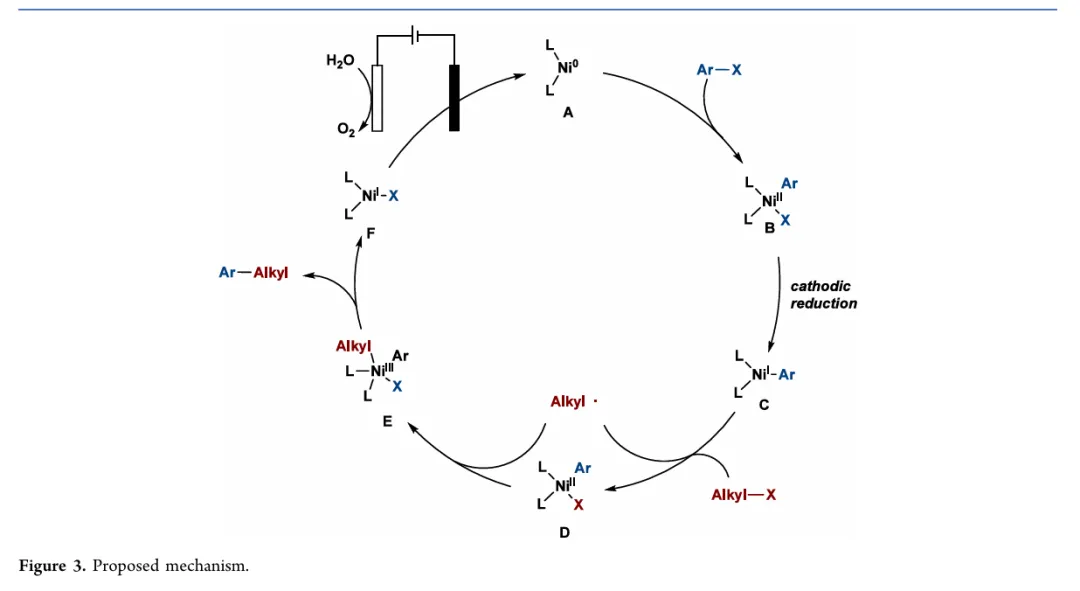 图8. 可能的反应机理 (图片来源于ACS Catal.)In summary, we have developed a novel nickel-catalyzed electroreductive cross-coupling method for C−C bond formation using H2O as an economical and sustainable sacrificial reductant. This electrochemical platform enables the versatile constructionof diverse C−C bonds, including Csp2−Csp3, Csp3−Csp3, and Csp−Csp3 linkages, from readily accessible aryl, alkenyl, alkynyl, and alkyl halide electrophiles, delivering products in yields of up to 99%. The approach eliminates the need for sacrificial metal anodes or stoichiometric organic reductants, offering a more sustainable and operationally straightforward alternative for cross-electrophile coupling. Furthermore, this electroreductive coupling strategy displays excellent functional group tolerance and is suitable for the late-stage derivatization of complex drugs and natural products. This operationally simple, electricity-driven method provides a sustainable and versatile platform for C−C bond formation. We anticipate that this strategy will find broad utility in synthetic and medicinal chemistry.
图8. 可能的反应机理 (图片来源于ACS Catal.)In summary, we have developed a novel nickel-catalyzed electroreductive cross-coupling method for C−C bond formation using H2O as an economical and sustainable sacrificial reductant. This electrochemical platform enables the versatile constructionof diverse C−C bonds, including Csp2−Csp3, Csp3−Csp3, and Csp−Csp3 linkages, from readily accessible aryl, alkenyl, alkynyl, and alkyl halide electrophiles, delivering products in yields of up to 99%. The approach eliminates the need for sacrificial metal anodes or stoichiometric organic reductants, offering a more sustainable and operationally straightforward alternative for cross-electrophile coupling. Furthermore, this electroreductive coupling strategy displays excellent functional group tolerance and is suitable for the late-stage derivatization of complex drugs and natural products. This operationally simple, electricity-driven method provides a sustainable and versatile platform for C−C bond formation. We anticipate that this strategy will find broad utility in synthetic and medicinal chemistry.