扬州大学王磊/张俊良(复旦大学)/南京大学王敏燕JACS:钯催化分子间炔丙基化反应对映选择性合成含季碳手性中心的醛类化合物
A highly efficient palladium-catalyzed enantioselective α-propargylation of α-alkyl-α-aryl disubstituted aldehydes with propargylic acetates has been developed using BINOL-derived chiral phosphoramidite ligands. This protocol affords a series of enantioenriched aldehydes bearing all-carbon quaternary stereocenters in moderate to good yields with high enantioselectivities. The reaction features a broad substrate scope, mild reaction conditions, facile scalability, and versatile downstream transformations. Density functional theory (DFT) calculations were conducted to elucidate the reaction mechanism and the origin of the enantioselectivity.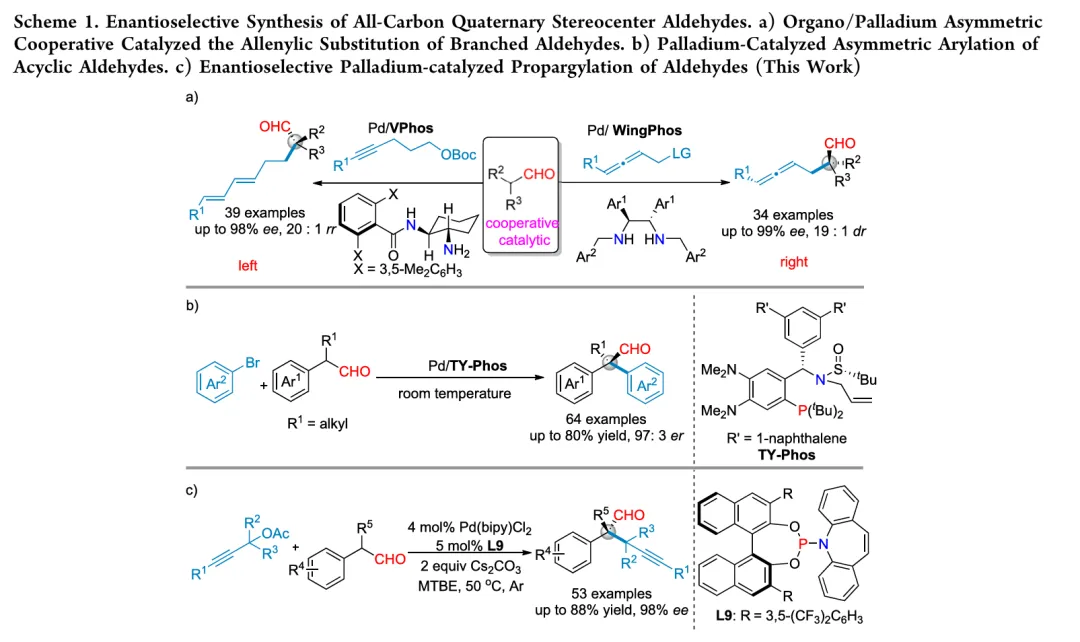
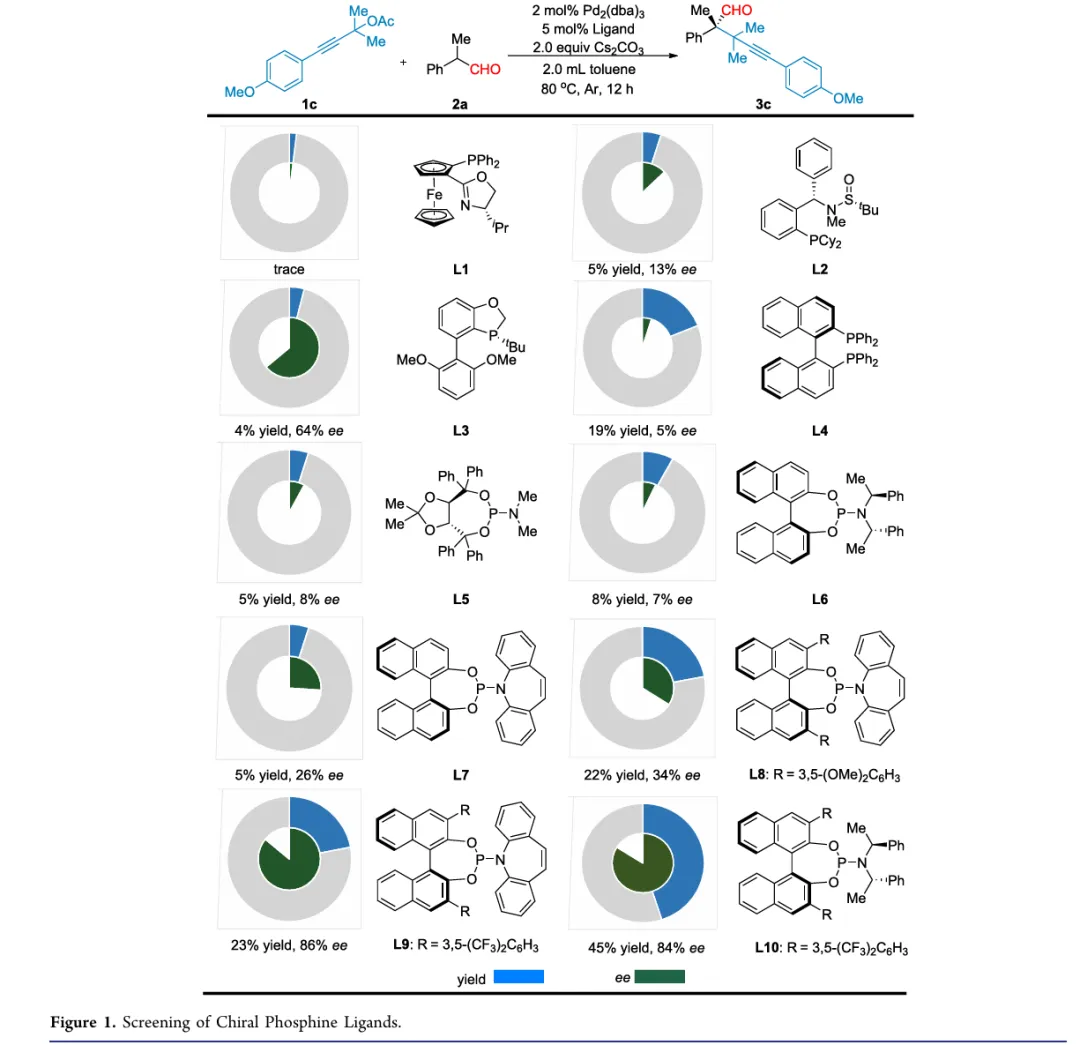
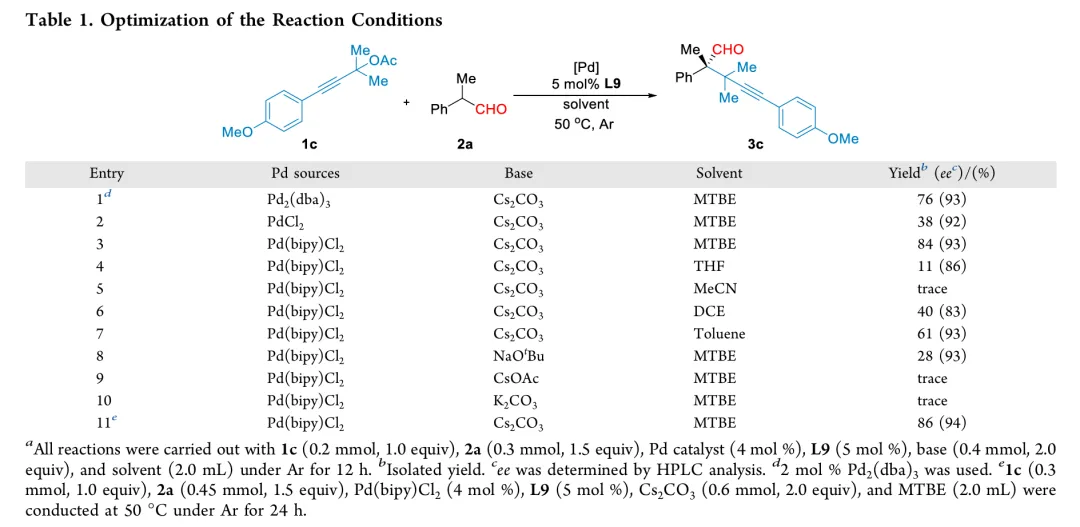 In conclusion, we have developed an efficient and enantioselective palladium-catalyzed α-propargylation of α-alkyl-α-aryl disubstituted aldehydes with propargylic acetates. This protocol provides straightforward access to enantioenriched aldehydes bearing all-carbon quaternary stereocenters in moderate to good yields with excellent enantioselectivities. Notably, a BINOL-derived phosphoramidite ligand plays a crucial role in achieving precise control over both chemoselectivity and enantioselectivity. The method features a broad substrate scope, mild reaction conditions, and operational simplicity, enabling gram-scale synthesis, as well as diverse downstream transformations to access structurally complex enantioenriched molecular frameworks. Density functional theory (DFT) calculations were conducted to elucidate the reaction mechanism, particularly the origin of enantiocontrol. Ongoing studies in our laboratory are focused on extending this strategy to other asymmetric palladium-catalyzed transformations.
In conclusion, we have developed an efficient and enantioselective palladium-catalyzed α-propargylation of α-alkyl-α-aryl disubstituted aldehydes with propargylic acetates. This protocol provides straightforward access to enantioenriched aldehydes bearing all-carbon quaternary stereocenters in moderate to good yields with excellent enantioselectivities. Notably, a BINOL-derived phosphoramidite ligand plays a crucial role in achieving precise control over both chemoselectivity and enantioselectivity. The method features a broad substrate scope, mild reaction conditions, and operational simplicity, enabling gram-scale synthesis, as well as diverse downstream transformations to access structurally complex enantioenriched molecular frameworks. Density functional theory (DFT) calculations were conducted to elucidate the reaction mechanism, particularly the origin of enantiocontrol. Ongoing studies in our laboratory are focused on extending this strategy to other asymmetric palladium-catalyzed transformations.